July 26, 2022 | Quality
How to Implement a QMS in Your Organization
Producing high-quality products your customers can depend on is the best way to achieve customer satisfaction and drive sales for manufacturing organizations. However, it takes robust, efficient, and sustainable quality management to deliver premium products consistently. To address these needs, manufacturers need to implement quality management systems (QMS).
This blog will cover the top questions surrounding quality management systems in manufacturing:
- What is a QMS in manufacturing?
- Why should you have a QMS?
- What are QMS documents?
- How can you create a QMS?
What is a Quality Management System (QMS)?
A quality management system (QMS) is a documentation system that controls product quality and manufacturing safety. Typically informed by regulatory standards, a QMS streamlines manufacturing, improves product quality, and enhances business growth and expansion.
By translating the requirements defined by regulatory authorities into security controls, a QMS helps organizations meet the needs of customers and streamline compliance.
Why Should Manufacturers Have a QMS?
As a manufacturer, you may be wondering, “Why have a QMS?” There are several reasons why QMS is critical in manufacturing. Most importantly, implementing an effective QMS helps:
- Protect your organization against potential litigation
- Establish and track operational goals
- Obtain customer feedback and improve overall satisfaction
- Streamline compliance documentation, assessment, and reporting
- Align employees with organization-specific manufacturing policies
- Disseminate information about critical processes and objectives across the organization
With a QMS in place, your organization will be better prepared to navigate and safeguard against potential quality assurance issues that arise during manufacturing.
What Are QMS Documents?
When developing a QMS, you must understand what the primary QMS documents are and how they work to streamline quality management. The four essential documents for a QMS are:
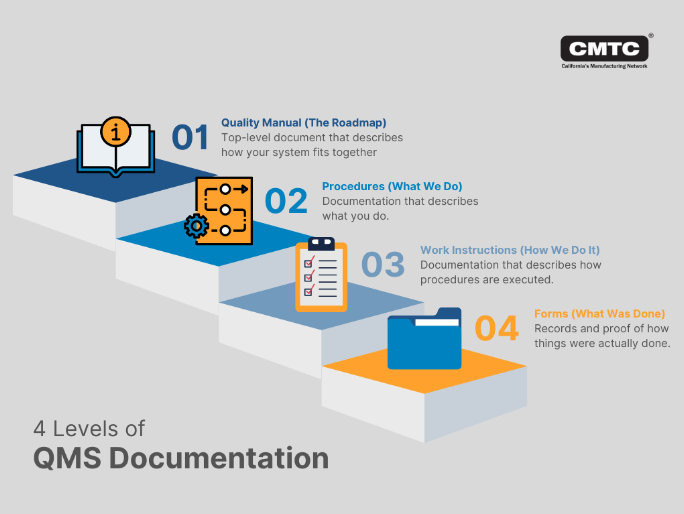
- Quality manual: Overseen by top-level management, a quality manual describes how a QMS fits together and governs the structure and implementation of a QMS.
- Procedures: Overseen by supervisor-level management, procedures describe the manufacturing processes that fall under the QMS and help customers understand the quality controls as well as provide top-level leadership with visibility into the QMS’ implementation.
- Work instructions: Owned by workers on the floor, work instructions are simplified descriptions of the procedures — typically including graphics for universal understanding — that employees use on a daily basis to do their jobs.
- Forms: As evidence of compliance with quality management standards, forms detail the who, what, and how of processes completed on a daily basis. They’re primarily used for regulatory audits but can also be used for internal quality assurance purposes.
Understanding QMS documentation will help simplify the steps to creating an effective QMS.
How to Create an Impactful QMS
Any organization looking to implement a QMS must develop and implement a plan that meets regulatory requirements and customer satisfaction standards at all stages of manufacturing.
Here are seven best practices for creating a QMS:
Before you start creating a QMS, it is crucial to consider your desired output. Defining your output will allow you to tailor the QMS to your specific manufacturing needs, as well as standards that regulate different manufacturing niches. For example:
- ISO 13485 caters to safety and quality in medical device manufacturing
- ISO 9001 guides general product quality management across industries
- AS9100 guides quality management in the aviation, space, and defense industries
- IATF 16949 caters to quality management in automotive manufacturing
Without a defined output, you won’t be able to create a QMS that fits your needs or allows you to qualify for critical industry certifications.
2. Consider Your Business’ Future Strategic Plans and Goals
With a determined output to guide the creation and implementation of a QMS, it’s also critical to align the QMS with your short- and long-term strategic plans. Thinking long-term will be helpful if your organization plans to expand into new industries or locations. You’ll need to account for regulatory requirements necessary at those thresholds and plan accordingly.
Two of the most critical considerations for creating and implementing a strategic QMS are:
- Global expansion: You must anticipate the regulatory requirements specific to your industry and location, as well as other markets and contexts you anticipate expanding into. Depending on your industry, compliance might involve frameworks such as ANVISA for medical device manufacturing in Japan, or Health Canada for consumer healthcare protection in Canada.
- Resource allocation: If you anticipate and incorporate future regulatory compliance requirements into a QMS proactively, you are less likely to incur the often significant resource costs of restructuring your entire QMS during global expansion.
When incorporated into quality management, strategic planning will help streamline regulatory compliance and expansion efforts across all stages of manufacturing.
3. Get Buy-In From Top-Level Leadership
When making decisions that impact quality management — whether in the short or long term — buy-in from top-level leadership is necessary to minimize gaps in the structure of your QMS.
Any discussions about QMS implementation must involve top-level leadership to ensure alignment between the organizational strategy and the execution of objectives. For example, it is critical for leadership to clarify expansion goals as far out as 10 or 15 years from the time a QMS is created. Guiding scope well in advance prevents errors and oversights down the line.
Discussions with top-level leadership around QMS development will help:
- Identify compliance requirements to include in the QMS
- Prevent wasteful allocation of resources during planning
- Streamline risk management processes relative to anticipated risks
- Simplify future manufacturing expansion efforts
- Avoid costs incurred in correcting QMS errors during audits
Top-level commitment is also critical when it comes to requesting additional resources for future quality management. Strong alignment between top-level leadership and designated quality management teams enables the development of a robust and functional QMS.
Once you have top-level leadership buy-in, it is also essential to document all decision-making processes and commitments in anticipation of any changes to top-level leadership. These could potentially undermine previous leadership commitments to the implementation of a QMS.
4. Create Documentation That Matches Your Processes
A QMS will provide little to no value if its stipulations do not match existing manufacturing processes one-to-one. It is all too common for companies to simply adopt the wording from standards and create QMS documentation that does not apply to any of their processes.
A versatile QMS with optimal documentation can be leveraged to:
- Boost productivity by generating higher manufacturing outputs
- Standardize processes to increase their reproducibility and safety
- Identify potential gaps in critical areas of the manufacturing pipeline
- Optimize employee training to achieve higher product quality
Importantly, QMS documentation should garner inputs from the owners of each process in the manufacturing pipeline. It’s a waste of stakeholders’ time and effort to document a QMS that doesn’t align with business processes as it will need to be heavily revised to capture any neglected details.
5. Internally Review and Externally Audit Your QMS to Certify
Another critical stage in QMS development is preparing for, conducting, and acting upon a QMS audit. Auditing is a way of validating your QMS and ensuring that it meets the manufacturing standards defined by regulatory authorities. Passing an audit is also a step toward achieving certification for applicable or mandatory regulatory standards.
The journey to QMS certification starts with an internal audit of your QMS documentation and the processes it supports. Here, you conduct an internal review of your quality management processes to identify gaps in compliance that should be reviewed by management.
When you feel prepared for the external audit, you can request one from a notified body.
External audits are usually conducted in two stages, depending on the notified body’s findings. Typically, you will get certified if there are no findings in the first audit. However, if the notified body identifies gaps in your QMS, then you must implement the recommended corrective actions prior to requesting a second audit. If that is successful, it will likely result in certification.
For proof of regulatory compliance, auditors usually request objective evidence that includes:
- Quality manuals detailing manufacturing protocols and respective controls
- Procedures followed by employees at each stage of manufacturing
- Forms and records that indicate process implementation on facility floors
Beyond evidence of compliance with your internal stipulations, employees may be asked to explain how they perform processes with respect to the documentation provided to auditors.
6. Implement the Processes You Outlined in Your System
Even after achieving certification, it remains critical that your manufacturing controls match the guidelines documented in your QMS. Constant engagement between quality management teams and manufacturing process owners will ensure that all relevant processes — including anticipated optimizations — are accurately captured in the QMS.
Furthermore, the descriptions of processes in the QMS should align with what is currently performed during each manufacturing process. Regardless of the quality of your manufacturing controls, any misalignment between the QMS documentation and existing processes could result in failed audits, delayed certification, and unnecessary remediation costs.
7. Practice Continuous Improvement
Like any other system with moving parts, a QMS must be subject to ongoing assessment and improvement to optimize its efficiency and adaptability, ultimately maximizing your profitability.
One of the biggest concerns with implementing a continuous quality improvement cycle is the potential effect on the overall quality system. However, this should not be an issue if you have a well-established foundation for a QMS. Proper structure from the outset should support:
- Seamless addition of new processes to drive profitability and sustainability
- Easy removal of redundancies such as obsolete compliance requirements
- Lean operations, powered by minimal waste (i.e., low-value outputs)
When built upon a solid foundation, continuous improvement will keep your QMS current, streamline regulatory compliance, and improve overall manufacturing efficiency.
CMTC’s Quality Management Services
At CMTC, we understand the path to achieving a robust, high-functioning QMS may not always seem straightforward, especially when it comes to developing QMS documentation. Quality concerns can impact a manufacturer’s reputation, customer satisfaction, and overall sales.
That’s where our quality management services come in. Our team of consultants draws on diverse expertise and skillsets to advise on all issues impacting California manufacturers.
CMTC will help you implement industry-specific best practices in your facility by leveraging our Production Effectiveness Solutions to expand the reach of your Quality Department. It doesn’t matter if you are a small or large business — CMTC can help! Contact us today to learn more.
About the Author
Gregg Profozich
Gregg Profozich is a manufacturing, operations and technology executive who believes that manufacturing is the key creator of wealth in the economy and that a strong manufacturing sector is critical to our nation’s prosperity and security now, and for future generations. Across his 20-year plus career in manufacturing, operations and technology consulting, Mr. Profozich helped manufacturing companies from the Fortune 500 to the small, independents significantly improve their productivity and competitiveness.
